RIQAS Standard Report
RIQAS Standard Report
There are a variety of RIQAS reports designed to enable quick and easy identification of any trends or test system issues. The standard quantitative report is provided in a user-friendly, one page per parameter format allowing a visual, at-a-glance assessment of performance. The standard quantitative report is split into several easy to interpret subsections each designed to save valuable laboratory time.
You can explore each of the report sections using the table below. Don’t forget, to enlarge the image, simply click on it.
RIQAS EQA Reports
RIQAS Reports Features
The text section provides a statistical breakdown of results by all methods, your method and instrument group. The Mean, CV% and Uncertainty or Measurement is presented for each comparison group.
Your laboratory’s result is compared to the Mean for Comparison (usually the instrument group Mean). Also included are the RIQAS performance indicators; SDI, Target Score and %Deviation.
Acceptable performance criteria:
- SDI <2
- Target Score >50
- %Deviation
The defined acceptable limits default to the RIQAS TDPA values but may be based on CLIA, biological variation or country specific limits.
Performance goals based on Biological Variation are also stated within the text section for information purposes.
The histogram chart provides an overview of how your laboratory’s result compares to the all method group, your method group and your instrument group. Your result is represented by a black triangle; the closer to the centre the better.
The chart is intended to provide a quick visualisation of performance compared to other method groups and can be used to identify any potential bias.
The Levey-Jennings chart plots the last 20 SDI’s and is extremely useful for monitoring EQA performance over time, allowing quick and easy identification of any trends or bias. The chart is colour coded making interpretation simple and easy; results that fall in the white area are excellent and those in the red area unacceptable.
The Target Score (TS) chart is a unique chart which displays your laboratory’s last 20 target scores delivering an instant, visual indication of performance. The TS chart is conveniently colour coded for even easier performance assessment, a TS >50 is acceptable. The TS is a numerical index relating your %Deviation from the mean to a Target Deviation for Performance Assessment (TDPA).
The %Deviation by sample chart displays the %Deviation for the last 20 EQA samples enabling identification of trends and shifts in performance. Similar to the other charts on the RIQAS report, the %Deviation by sample chart is shaded to indicate the limit of acceptable performance. A black dot within the white section of the chart will represent results with a %Deviation within your acceptable limits of performance; a black dot within the red section of the chart will represent results with a %Deviation outside your acceptable limits of performance. %Deviations are not influenced by the performance of your peers, as seen with the standard deviation index (SDI) and therefore is a better indicator of individual performance.
The %Deviation by concentration chart enables easy detection of possible concentration related biases. Unlike the other charts provided on the report, the %Deviation by concentration chart displays the concentration range of the previous 20 samples along the bottom of the chart.
Using this scale along with the percentage deviation, you are provided with a rapid assessment of your %Deviation in relation to the concentration of the:
• Current sample (represented by a square)
• Your 19 previous results (represented by circles)
This chart provides an easy interpretation of potential positive or negative biases at high or low concentrations, or whether a particular sample is a random outlier.
Located at the back of the standard quantitative report, our quick reference summary page details the performance for each registered parameter in the programme.
Within the performance column, RIQAS provides an effortless method of assessing the performance of each parameter within the sample distribution. When a red triangle appears next to the parameter, this indicates that all performance indicators (SDI, TS and %DEV) have exceeded the performance criteria.
The performance indicator limits for each parameter are exceeded when your result produces:
• An SDI greater than +/- 2 standard deviations.
• A Target Score less than 50 (only when Target Scoring is available)
• A %Deviation greater than your set acceptable limits of performance.
A summary CSV file is available on request to all participating laboratories. The report provides a summary of all statistics, acceptable limits and performance indicators as a .csv file for each sample in the cycle.
A retrospective statistics summary is also available, four weeks after the final submission date for parameters where a result was not submitted on time.
RIQAS Parameters List
RIQAS is the largest international External Quality Assessment (EQA)/ Proficiency Testing (PT) scheme, there are currently more than 76,000 participants in 139 countries. World renowned for reducing the number of individual programmes required by even the most demanding laboratories, RIQAS covers 360 parameters across 36 flexible multi-parameter programmes. Effective consolidation in this way will not only deliver real cost savings but free up storage space and ultimately reduce the time spent preparing multiple samples at each survey.
RIQAS Parameter List
1-25-(OH)2-Vitamin D
17-OH-Progesterone
25-OH-Vitamin D
5-HIAA
α-1-Acid Glycoprotein
α-1-Antitrypsin
α-2-Macroglobulin
ABO Group
ACE
Acid Phosphatase (Prostatic)
Acid Phosphatase (Total)
ACR
ACTH
AFP
Albumin
Albumin/Microalbumin
Aldosterone
Alkaline Phosphatase
ALP
ALT
ALT (ALAT)
AMH
Amikacin
Ammonia
Amylase
Amylase (Pancreatic)
Amylase (Total)
Androstenedione
Anti Streptolysin O
Antibody Identification
Antibody Screen
Anti-CMV (Total)
Anti-CMV IgG
Anti-CMV IgM
Anti-EBNA IgG
Anti-EBV VCA IgG
Anti-EBV VCA IgM
Antigen Typing
Anti-HAV (Total)
Anti-HAV IgM
Anti-HBc
Anti-HBc IgM
Anti-HBe (Total)
Anti-HBs (Total)
Anti-HCV
Anti-HIV combined
Anti-HIV-1
Anti-HIV-2
Anti-HSV1 IgG
Anti-HSV1 IgM
Anti-HSV1/2 IgG
Anti-HSV1/2 IgM
Anti-HSV2 IgG
Anti-HSV2 IgM
Anti-HTLV combined
Anti-HTLV I
Anti-HTLV II
Anti-Measles IgG
Antimicrobial Susceptibility Testing
Anti-Mumps IgG
Anti-Rubella IgG
Anti-Rubella IgM
Anti-Tg
Antithrombin III
Anti-Toxoplasma IgG
Anti-Toxoplasma IgM
Anti-TPO
Anti-TSH Receptor (TRAb)
Anti-VZV IgG
Apolipoprotein A1
Apolipoprotein B
aPTT
AST
AST (ASAT)
β-2-Microglobulin
Benzoylegonine
Bicarbonate
Bile Acids
Bilirubin
Bilirubin (Direct)
Bilirubin (Total)
Blood
BNP
Buprenorphine
Ca++
CA125
CA15-3
CA19-9
Caffeine
Calcitonin
Calcium
Calcium (Ionised)
Calcium, Adjusted
Cannabinoids (THC)
Carbamazepine
Carboxyhaemoglobin (COHb / HbCO)
CEA
Ceruloplasmin
Chloride
Cholesterol
Cholesterol (Total)
Cholinesterase
Ciclosporin
CK NAC
CK, Total
CK, total (CPK)
CK-MB (Activity)
CK-MB (Mass)
Cl-
CO2(Total)
Compatability (Cross-Match)
Complement C3
Complement C4
Conductivity
Copper
Cortisol
Cotinine
C-Peptide
C-Reactive Protein
Creatinine
CYFRA 21-1 (Cytokeratin 19 fragment)
D-3-Hydroxybutyrate
d-Amphetamine
D-dimer
Deoxyhaemoglobin (RHb / HHb)
DHEA (Unconjugated)
DHEA-S
Digoxin
Direct Bilirubin
d-Methamphetamine
Dopamine
EDDP
eGFR (estimated glomerular filtration rate)
Epidermal Growth Factor (EGF)
Epinephrine
ESR
Estriol
Ethanol
Ethosuximide
Everolimus
Factor II
Factor IX
Factor V
Factor VII
Factor VIII
Factor X
Factor XI
Factor XII
Ferritin
Fibrinogen
Folate
Free Morphine
free β-hCG
Fructosamine
FSH
Galactose
Gastrin
Gentamicin
GGT
GH
GLDH
Glucose
γGT
Haematocrit (HCT)
Haemoglobin (Hb)
Haemolysis
Haptoglobin
HbA1c
HBDH
HBsAg
hCG
HDL
HDL-Cholesterol
Homocysteine
hsCRP
Icteric
IgA
IgE
IGF-1
IgG
IgM
Inhibin A
Insulin
Interferon gamma (IFN-y)
Interleukin-1 alpha (IL-1α)
Interleukin-1 beta (IL-1β)
Interleukin-10 (IL-10)
Interleukin-2 (IL-2)
Interleukin-4 (IL-4)
Interleukin-6 (IL-6)
Interleukin-8 (IL-8)
Iron
K+
Kappa Light Chain (Free)
Kappa Light Chain (Total)
Ketones
Lactate
Lambda Light Chain (Free)
Lambda Light Chain (Total)
LD (LDH)
LDH
LDL-Cholesterol
Leukocytes
LH
Lipaemic
Lipase
Lipoprotein (a)
Lithium
Lorazepam
LSD
Magnesium
MDMA
Mean Cell Haemoglobin (MCH)
Mean Cell Haemoglobin Concentration (MCHC)
Mean Cell Volume (MCV)
Mean Platelet Volume (MPV)
Metanephrine
Methadone
Methaemoglobin (MetHb)
Methotrexate
Monocyte Chemottractant Protein-1 (MCP-1)
Myoglobin
Na+
NEFA
Nitrite
Non-HDL Cholesterol
Norepinephrine
Normetanephrine
Norpropoxyphene
Nortryptyline
NT proBNP
Oestradiol
Osmolality
Osteocalcin
Oxalate
Oxazepam
Oxygen Content (O2CT)
Oxygen Saturation (sO2 / Vol O2)
Oxyhaemoglobin (O2Hb / HbO2)
P24
PAPP-A
Paracetamol
pCO2
pH
Phencyclidine
Phenobarbital
Phenytoin
Phosphate
Phosphate (Inorganic)
Plasma Renin Activity
Plasminogen
Plateletcrit (PCT)
Platelets (PLT)
PlGF
pO2
Potassium
Prealbumin (Transthyretin)
Primidone
Procalcitonin
Progesterone
Prolactin
Protein
Protein (Total)
Protein C
Protein S
PSA
PSA (Free)
PSA (Total)
PT (including INR)
PTH
Red Blood Cell Count (RBC)
Red Cell Distribution Width (RDW)
Renin (Direct Concentration)
Retinol Binding Protein
Rh(D)
Rheumatoid Factor
Salicylate
Salicylic Acid
Secobarbital
sFlt-1
sFlt-1/PlGF ratio
SHBG
Sirolimus
Sodium
Specific Gravity
Strain Identification
Syphilis
T3 (Free)
T3 (Total)
T4 (Free)
T4 (Total)
Tacrolimus
Testosterone (Free)
Testosterone (Total)
Theophylline
Thyroglobulin
TIBC
Tobramycin
Total Bilirubin
Total Haemoglobin
Total Haemoglobin (tHb)
Total hCG
Total White Blood Cell Count (WBC)
Transferrin
Triglycerides
Troponin I
Troponin T
Trypanosoma Cruzi Antibodies
TSH
TT
Tumour Necrosis Factor alpha (TNF-α)
UIBC
Unconjugated Oestriol
Urea
Uric Acid
Urobilinogen
Valproic Acid
Vancomycin
Vascular Endothelial Growth Factor (VEGF)
Vitamin B12
VMA
What is Measurement of Uncertainty?
Measurement Uncertainty (MU) relates to the margin of doubt that exists for the result of any measurement, as well as how significant the doubt is. For example, a piece of string may measure 20 cm plus or minus 1 cm, at the 95% confidence level. As a result, this could be written: 20 cm ±1 cm, with a confidence of 95%. Therefore, we are 95% sure that the piece of string is between 19 cm and 21 cm long.
Standards such as ISO 15189 require that the laboratory must determine uncertainty for each test. However, they have not specified how this should be done.
How do we calculate Measurement Uncertainty using QC data?
Employing your QC data to calculate uncertainty makes several assumptions; your test system is under control, the patient samples are treated in the same manner as your controls and gross outliers have been removed. If you choose to use your QC data to calculate this you should ensure that you use a commutable control with a matrix similar to that of a patient sample, with analytes present at clinically relevant levels
To calculate MU, labs must look at the intra-assay precision and inter-assay precision of their test.
Intra-assay precision: Sometimes known as ‘within run’ precision, is where 20 or more replicates of the same sample are run at the same time, under the same conditions (calculated from a single experiment). Intra-assay precision helps to assess systematic uncertainties
Inter-assay precision: Sometimes known as ‘between run’ precision, is where 20 or more replicates are run at different times – e.g. 1 replicate every day for 20 days (can be calculated from routine IQC data). Inter-assay precision can help identify random uncertainties within the test system.
*The Australian Association of Clinical Biochemists (AACB) recommends that at least 6 months’ worth of QC data are used when calculating the inter-assay precision1.
Once the data is collected, you must calculate the standard error of the mean (SEM) of the intra-assay precision (A) and the SD of the inter-assay precision (B) in order to measure the uncertainty (u). Once A and B have been calculated, they need to be squared, added together and the square root of the sum found:
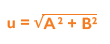
As uncertainty is calculated as SD and 1SD is equal to 68% confidence on a standard Gaussian curve, we can conclude that if we multiply using a coverage factor of 2, we can attain 2SD confidence of 95%. This is known as the Expanded Uncertainty (U):
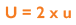
What is the Advantage of Measurement Uncertainty for a lab?
Labs need to carry out MU as it is a requirement of ISO 15189. It states: “The laboratory shall determine measurement uncertainty for each measurement procedure, in the examination phases used to report measured quantity values on patients’ samples. The laboratory shall define the performance requirements for the measurement uncertainty of each measurement procedure and regularly review estimates of measurement uncertainty”.
MU also helps determine whether the difference between two results is negligible due to uncertainty or significant due to a genuine change in condition of the patient; giving labs a greater confidence in reported results.
How can Randox help?
Our new Acusera 24.7 Live Online software provides automatic calculation of MU, saving valuable time and helping labs meet ISO 15189 requirements with ease.
Contact marketing@randox.com to find out how your lab can benefit from Acusera 24.7 Live Online
Take steps to prevent incorrect patient results by making one simple change
According to the NHS Litigation Authority; in 2015 within the UK alone, £193,680,744.30 was spent on ‘wrong diagnosis’ or ‘failed/delayed diagnosis’ causing huge financial strain and impact on labs.
With approximately 75% of clinical decisions and diagnosis based on laboratory test results. The only way to guarantee a high degree of accuracy is to implement a good Quality Control plan. The importance of this is recognised globally, several bodies exist internationally including ISO (International organisation for standardisation) who have developed a set of guidelines and quality systems to ensure the reliability of laboratory test results.
So what can you do to improve accuracy and reliability?
Choose a third party QC
ISO 151589:2012 Section 5.6.2.2 states that “the use of third party control materials should be considered, either instead of, or in addition to, any control materials supplied by the reagent or instrument manufacturer”.
First Party Controls are those manufactured by the instrument/reagent manufacturer. These controls are optimised specifically for use with the manufacturers test system and therefore will mask a multitude of weaknesses. First Party Controls tend to result in perceived accuracy and a biased assessment of performance.
Third Party Controls on the other hand are designed to be completely independent and are not optimised for use with a specific test or system. Leading manufacturers of third party controls will assign target values based on data collected from thousands of independent laboratories, ensuring the availability of statistically robust multi-method, multi-analyser data. Therefore laboratories using Third Party Controls can be assured of unbiased error detection across multiple platforms.
Randox Acusera is a world leading manufacturer of true third party controls providing a cost effective, high quality solution for any laboratory-regardless of size or budget.
Look out for QC samples with clinically relevant concentrations
ISO 15189:2012 states that ‘The laboratory should choose concentrations of control materials wherever possible, especially at or near clinical decision values, which ensure the validity of decisions made’.
It is important to assess the full clinical range of an assay i.e. the range between the lowest and highest results which can be reliably reported. In order to make sure a laboratory instrument is performing accurately across the full clinical range and in particular at the medical decision level, QC materials that cover low, normal and elevated concentrations should be used.
Due to the superior manufacturing process used by Randox, QC target values consistently cover the MDL of tests. By ensuring the controls in use cover clinical decision levels laboratories can be confident of the reliability and accuracy of the patient results they release.
Opt for a commutable control material
A good QC material has many essential properties but above all, controls must perform consistently and reflect the performance of patient samples – if a control meets these requirements then we can say it is commutable. Having a commutable control would aid in the prevention of incorrect patient results because they replicate the performance of a patient sample and react to the test system in a similar manner. Use of a commutable control will also reduce costly shifts in QC target values when reagent batch is changed.
At Randox we take quality seriously, that’s why all QC products are manufactured to the highest possible standard, delivering controls of unrivalled quality. Designed to be commutable, the Acusera range will ensure accurate and reliable instrument performance while simultaneously helping laboratories to meet ISO 15189:2012 requirements. A good QC process will include the use of Third Party Controls, Clinically Relevant Concentrations and controls which can be described as commutable. By employing Quality Control’s that encompass these traits, a laboratory professional can be certain that they have taken the necessary steps to decrease incorrect results and therefore potential misdiagnosis.












